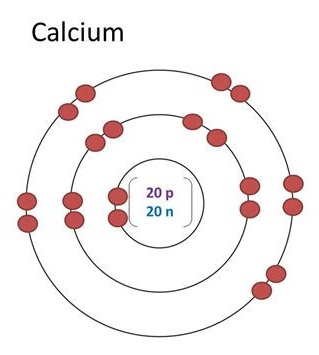
It is because of this order in which the atomic orbitals are filled as stated in Afbau’s principle that the electronic configuration of calcium is 2, 8, 8, 2 neither 2, 8, 9, 1 nor 2, 8, 10. Therefore, number of electrons in calcium = 20. So the number of valence electrons in Ca is 2. There are two valence electrons in the outermost shell. Here, the number of valence electrons in calcium is 2. The valency of a group 2 element is decided by the number of valence electrons. What is the valence of Ca?Ĭalcium is in group 2. In order to write the Calcium electron configuration we first need to know the number of electrons for the Ca atom (there are 20 electrons ). The most common form of calcium has 20 protons, 20 neutrons, and 20 electrons. How many core and valence electrons are in Ca?Ĭa has 18 core electrons and 2 valence electrons. Hence, Ca+ does not have eight electrons in its valence shell. ✏️ Exercise \(\PageIndex \) Tell which group of elements (alkali metals, alkaline earth metals, halogens, noble gases) has atoms with the specified number of valence electrons?Īnswer A alkali metals Answer B alkaline earth metals Answer C halogens Answer D noble gases Does Ca+ have 8 valence electrons? Aluminum (Al) is located in Group IIIA (Group 13), so it has 3 valence electrons.Potassium (K) is located in Group IA (Group 1), so it has 1 valence electron.As noted above, helium is the only exception for the main group elements. However, one atom only has two electrons, so it could never have more than 2 valence electrons. Helium (He) is located in Group VIIIA (Group 18).Sulfur (S) is located in Group VIA (Group 16), so it has 6 valence electrons.An interactive periodic table may be found here, ✅ Example \(\PageIndex \): Number of Valence Electrons How many valence electrons are in one atom of each element? Figure \(\PageIndex \): The modern version of the periodic table. Transition elements (groups designated with a Roman numeral followed by the letter B) usually have two valence electrons, though we will not be working with valence electrons for the transition elements in this text. In other words, the maximum number of valence electrons that an atom may have is eight. When the outermost energy level is any other level beyond the first energy level, it may contain up to eight valence electrons.Since helium atoms only have two electrons and the outermost energy level is the first energy level, there can only be two valence electrons.The first energy level holds a maximum of two valence electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
